
The ICHOM Set of Patient-Centered Outcome Measures for Non-metastatic Breast cancer is the result of hard work by a group of leading physicians, measurement experts and patients. It is our recommendation of the outcomes that matter most to persons with Breast cancer. We urge all providers around the world to start measuring these outcomes to better understand how to improve the lives of their patients.
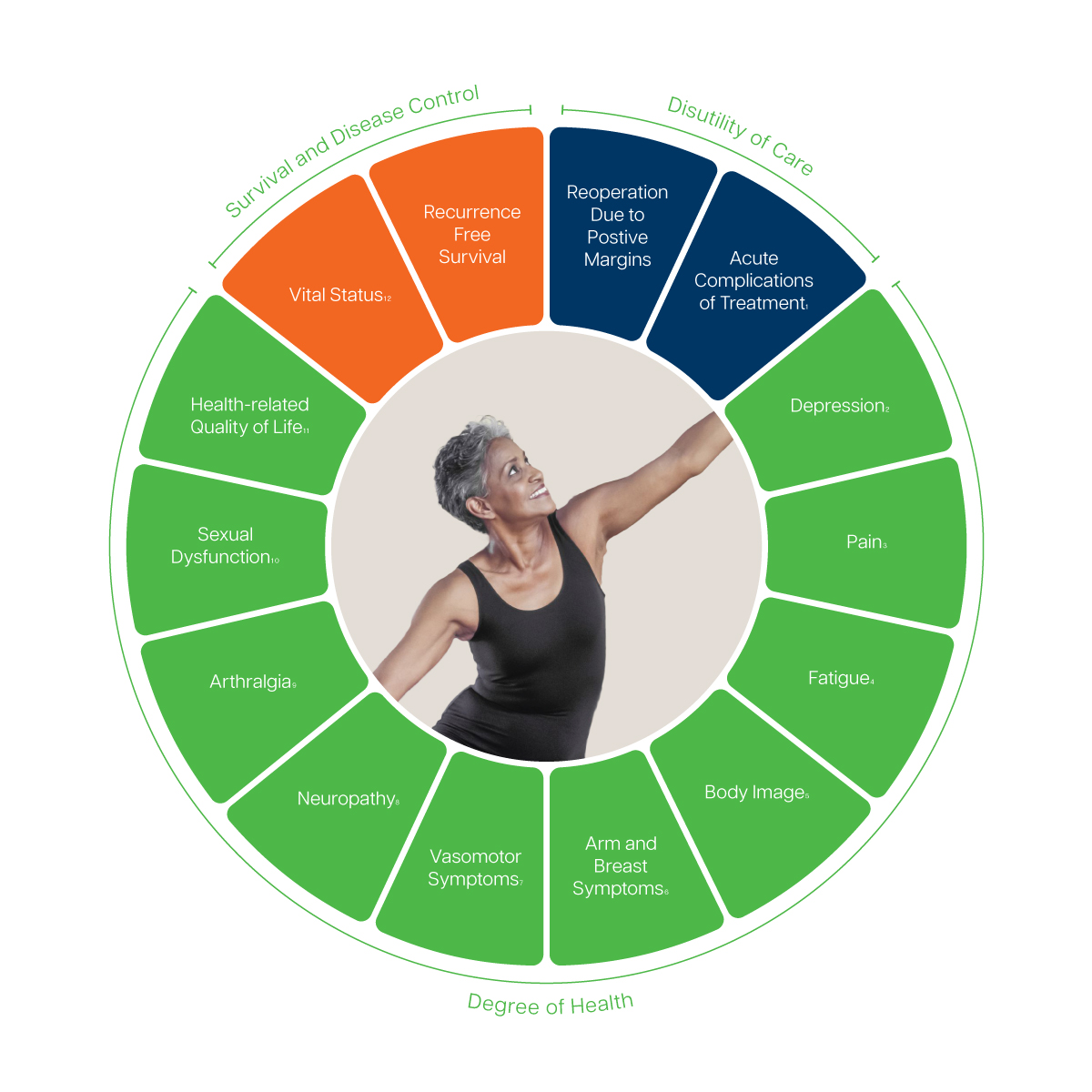
1 Complications will be recorded based on the type of therapy needed or action required to correct the complication as described in the Clavien-Dindo Classification and CTCAE v4.0
2 Includes depression and anxiety.
3,4 Recommended to track via the EORTC Quality of Life Questionnaire – Core Questionnaire (EORTC QLQ-C30).
5 Includes body image and satisfaction with breast(s). Recommended to track via the EORTC Quality of Life Questionnaire – Breast Cancer Specific Questionnaire (EORTC QLQ-BR45) and the BREAST-Q 2.0 – Satisfaction with Breasts.
6 Includes arm and breast symptoms. Recommended to track via the EORTC Quality of Life Questionnaire – Breast Cancer Specific Questionnaire (EORTC QLQ-BR45).
7, 8, 9 Recommended to track via the EORTC Quality of Life Questionnaire – Breast Cancer Specific Questionnaire (EORTC QLQ-BR45).
10 Includes sexual functioning and vaginal symptoms. Recommended to track via the EORTC Quality of Life Questionnaire – Breast Cancer Specific Questionnaire (EORTC QLQ-BR45).
11 Includes physical, emotional, cognitive and social functioning, ability to work and overall well-being. Recommended to track via the EORTC Quality of Life Questionnaire – Core Questionnaire (EORTC QLQ-C30).
12 Includes overall and cause-specific survival.
Team that developed this set
AUSTRALIA
Rodney Cooter | Monash University
Geoff Delaney | South Western Sydney, Local Health District
Patricia Hancock* | Breast Cancer Network Australia
Wee Loon Ong | Peter MacCallum Cancer Centre
Christobel Saunders | University of Western Austrlia
Lisa Sheeran | Peter MacCallum Cancer Centre
BELGIUM
Francois Duhoux | Cliniques Universitaires Saint-Luc
IRELAND
John Browne | University College Cork
MALAYSIA
Cheng Har Yip | Subang Jaya Medical
ITALY
Karen Benn* | Europa Donna
MEXICO
Felicia Knaul* | Cancer de Mama
NETHERLANDS
Anne Knip* | Borstkanker Vereniging Nederland
Linetta Koppert | Erasmus MC Cancer Institute
Marc Mureau | Erasmus MC Cancer Institute
Mark Stoutjesdijk | Ikazia Hospital Rotterdam
Marie-Jeanne Vrancken Peeters | Antoni van Leeuwenhoek
SWEDEN
Yvonne Wengström | Karolinska Institutet
UNITED STATES
Kimberly Allison | Stanford University, School of Medicine
Patricia Ganz | UCLA Schools of Medicine & Public Health
Reshma Jagsi | University of Michigan
Henry Kuerer | MD Anderson Cancer Center
Sarah McLaughin | Mayo Clinic Jacksonville
Ann Partridge | Dana-Farber Cancer Institute
Dereesa Reid* | Hoag Orthopedic Institute
Thomas Smith | Johns Hopkins Institute
*Patient representative
Are you implementing ICHOM Sets?
If your are implementing ICHOM Sets, please help us understand more about your journey by filling in our Implementation Survey. Click on the link below to complete:
Implementation Map
We would like to add you to our Implementation Map if you are implementing or have implemented ICHOM Sets. Please click on the button below for more information.






















