The ICHOM Set of Patient-Centered Outcome Measures for Epilepsy is the result of hard work by a group of leading physicians, measurement experts and patients. It is our recommendation of the outcomes that matter most to patients with Epilepsy. We urge all providers around the world to start measuring these outcomes to better understand how to improve the lives of their patients.
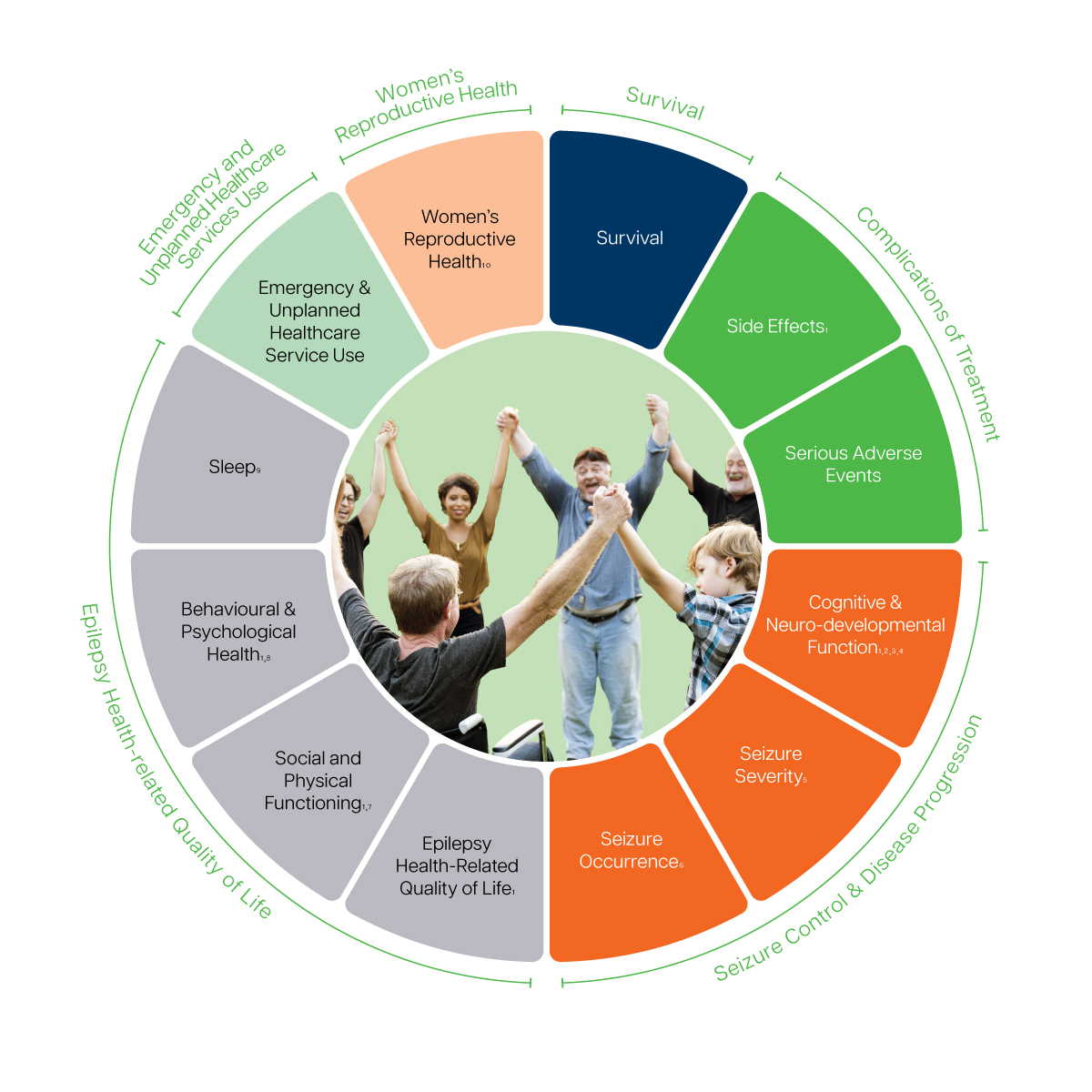
- Epilepsy health-related quality of life reported with QOLIE-10 in adults, and QOLCE-16 (parent-report) or CHEQOL-25 (self-report) in children and adolescents.
- Including Behavioural development, Motor development, and Cognitive development (pediatrics only), & Attention/Inattention and Memory Impairment (all patients).
- Memory Impairment & Attention/Inattention reported with the PROMIS v2.0 Cognitive Function SF 4a in adults, and CHEQOL-25 in children and adolescents.
- Behavioural development, Motor development, and Cognitive development measured using the mCHAT (16-48 months) or national assessment methods (>48 months).
- Including Prolonged/recurrent clustered seizures requiring emergency treatment, Seizure-related injury and Incontinence. Optional patient-report with the LSSS 2.0 in adults.
- Including seizure type, Seizure frequency, Seizure freedom, Emergence of new seizure type
- Including impact of epilepsy on education/work and daily life activities
- Depression and Suicidality reported with the PHQ-9 in adults. Depression measured with the PROMIS Pediatric Item Bank v2.0 – Depressive Symptoms/Parent Proxy Item Bank/ Early Childhood Parent Report SF 4a in children and adolescents. Suicidality in children and adolescents reported with the ASQ. Anxiety reported with the GAD-2 in adults, and PROMIS Anxiety Item Bank SF 8a/Parent proxy SF 8a/Early Childhood Parent Report SF 4a in children and adolescents.
- Including Sleep Adequacy. Reported by the PROMIS Sleep Disturbance 4a in adults, and the PROMIS Pediatric Pediatric Sleep Disturbance 4a in children and adolescents.
- Including pregnancy and delivery complications, and congenital malformations in offspring. Only measured in women of a reproductive age.
ICHOM Pediatric and Adult Epilepsy Project Team:
- Working Group Chair: Prof. Anne Berg – Northwestern University
- Working Group Research Fellow: Dr. James Mitchell – University of Liverpool
- Project Manager: Frieda Sossi – ICHOM, UK
- Research Associate: Isabel Miller – ICHOM, UK
- Research Associate: Dr. Paula Blancarte Jaber – ICHOM, UK
- Director of Outcomes Research: Dr. Zofia Das Gupta – ICHOM, UK
- Director of Outcomes Research: Dr. Luz Fialho – ICHOM, UK

Team that developed this set
Australia
Christian Gericke | The University of Queensland, Brisbane, Australia
Emilio Perucca | Department of Medicine (Austin Health), The University of Melbourne, and Department of Neuroscience, Central Clinical School, Monash University, Melbourne, Australia
Austria
Tim von Oertzen | Department of Neurology, Kepler University Hospital, Linz, Austria
Canada
Gabriel Ronen | McMaster University, department of Pediatrics, andCanChild Centre for Childhood Disability Research, Hamilton, Ontario, Canada
Cassidy Megan* | Founder of Purple Day
Chile
Tomás Mesa | Universidad Católica de Chile. IBE Chilean Chapter
Germany
Leonie Wollscheid*
India
Mamta Bhushan Singh | All India Institute of Medical Sciences, New Delhi, India
Ireland
Lorraine Lally*
Israel
Bruria Ben Zeev | Safra pediatric hospital, Sheba Medical Centre ,Sackler School of Medicine Tel Aviv University
Italy
Francesca Sofia* | International Bureau for Epilepsy
Kenya
Sharon Mbugua* | Mental Health Alliance of Kenya
Malawi
Action Amos* | International Bureau of Epilepsy/University of Edinburgh
Mexico
Lilia Nuñez-Orozco | Nacional Medical Center “20 De Noviembre, Mexico City
Singapore
Derrick Wei Shih Chan | KK Women’s and Children’s Hospital
South Africa
Jo M Wilmshurst | University of Cape Town
Sharlene Andrea Cassel* | Epilepsy SA
Sweden
John Eric Chaplin | Sahlgrenska Academy at Gothenburg University
UK
Angela Audrey Pullen* | Formerly Epilepsy Action, Employed by Epilepsy Research UK Board Director HealthWatch Leeds
Gus Baker | University of Liverpool
Helen Cross | UCL GOS Institute of Child Health
USA
Aatif M. Husain | Duke University Medical Center
Jeffrey Bolton | Boston Children’s Hospital; Harvard Medical School
Joan K Austin | Indiana University School of Nursing
Martha Sajatovic | Case Western Reserve University
Scott Badzik*
* Patient Representative
Are you implementing ICHOM Sets?
If your are implementing ICHOM Sets, please help us understand more about your journey by filling in our Implementation Survey. Click on the link below to complete:
Implementation Map
We would like to add you to our Implementation Map if you are implementing or have implemented ICHOM Sets. Please click on the button below for more information.







































